Hello and welcome to this design properties for plastics engineering post. As in this previous post given as an outlook, we continue today with blends of Polycarbonate with styrene and polyester polymers.
What are polymer blends?
Blends are made by mixing a polymer A with a polymer B. In case we can mix both polymers we will create a miscible blend with one glass transition temperature (Tg). In case the two polymers cannot be easily combined, an immiscible blend is created having two glass transition temperatures. Examples of blends are PC/ABS, PC/ASA, PC/PBT, PC/AES, PPE/PS/PA, and PA/PPA.
Why are polymer blends useful?
Polymer blends help to overcome the lack of some properties (flexibility, transparency, low density,...) which is needed for the application. They help to fulfil the product requirements. By blending of expensive polymers, lower overall material costs by a slightly changed property profile can be achieved too.
Important properties of blends for part design - example impact performance
Polycarbonate shows excellent impact performance from room temperature up to 40°C (Figure 1). Below 0°C PC shows brittle behaviour and it shows that temperature influences the impact behaviour of polymers. The transition from brittle to ductile behaviour is very sudden. To avoid such a rapid transition, blending with ABS can be done. This allows it to have a more homogeneous behaviour at lower and higher temperatures.
 |
| Figure 1: comparison of Izod Notched Impact test of PC, PC/ABS, and ABS. |
Important properties of blends for part design - example chemical resistance
Another example is the blend out of PC and the polyester PBT. Thoroughness and stability is offered by PC, however chemical resistance, especially towards solvents is limited. This is where PBT helps and provides enhanced solvent resistance. Using a PC/PBT blend in your application will result in a product which has good heat, chemical, and impact resistance. It covers impact performance at high temperatures as well as chemical resistance allowing this blend to be used for example as gear casing material in automotive.
Important properties of blends for part design - example increase flow for better part filling
Alloying amorphous polymers with semi-crystalline polymers increases also the flow rate which is an advantage during processing. A Polycarbonate has usually a melt flow of 10 g/10 minutes. If a blend in a ABS, flow rate can be increased to 15 g/10 min and by using a PBT it can be doubled to 20 g/10 minutes.
Thermal-mechanical properties of blends: Dynamic Mechanical Analysis (DMA) of PC-blends
Figure 2 shows the DMA curves of ABS (Cycolac T), PC/ABS (Bayblend FR110), PC/PBT Xenoy 6123), PC/PBT with 10 wt% glass fiber (Xenoy 6240), and PC/PET (Makroblend UT1018). Advantages of using PC/ABS instead of PC alone is the enhanced processability and uniform impact behaviours (low and high temperature almost same). Using PC/PBT will enable applications where higher temperature performance, together with chemical resistance is needed. If higher mechanical properties are needed too, then adding 10 wt% glass fiber as reinforcement is enough to double the modulus of the blend over the temperature range. Furthermore, PC/PBT has a single glass transition temperature which is around 127°C, indicating the miscibility of the two polymers in the amorphous phase.
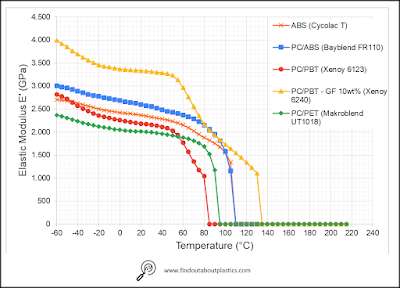 |
| Figure 2: DMA of ABS, PC/ABS, PC/PBT, and PC/PET [5]. |
What other important PC-blends are available?
Apart from the aforementioned blends PC/ABS and PC/PBT, there are some more interesting PC blends such as PC/ASA and PC/AES:
PC/ASA: great temperature stability, together with high UV resistance and can withstand cold temperatures without cracking.
PC/AES: combining the strength, toughness, and heat resistance of Polycarbonate with the stress cracking and weather resistance of AES. Suitable for outdoor applications, building materials, and automotive components.
PBT/PET: in PBT/PET blends, PET primarily contributes to cost-effectiveness and plays a role in enhancing the mechanical properties of the blend. PBT is more expensive than PET, and its inclusion in the blend makes the composite more cost-competitive. PET also influences the overall stiffness and strength of the PBT/PET blend.
PET/ASA: blending PET with ASA is enhancing weather resistance, UV resistance, and improving the thermal stability of the blend.
Table 1 compares major properties of PC/ASA and PC/AES to PC/ABS and PC/PBT.
Table 1: Performance overview of PC blends [4].
| PC Blend |
Tensile strength at yield (MPa) |
Tensile elongation at break |
Density (g/cm3) |
Heat deflection temperature at 1.8 MPa (°C) |
Notched Izod impact (J/m) |
Water adsorption (%) |
| PC/ASA (EnVoy 2506UR) |
62 |
25 |
1.17 |
105 |
481 |
0.5 |
| PC/AES (EnVelop 1812) |
49 |
>60 |
1.12 |
96 |
641 |
0.25 |
| PC/PBT (EnCounter F5PC/PBT10GF) |
67 |
/ |
1.3 |
101 |
61 |
0.30 |
| PC/ABS (EnValoy F0PC/ABS) |
61 |
40 |
1.18 |
94 |
534 |
0.30 |
Conclusions
Blending PCs with different styrene polymers or polyesters help to overcome different property gaps in an efficient way. PC blends represent an important addition to the material selection toolbox which you can try out during your next project.
If you have interest in testing PC blends and need some sample materials you can let me know here.
Thanks and #findoutaboutplastics
Greetings,
Herwig Juster
[1] https://www.youtube.com/watch?v=QrbHcEagUZM
[2] http://www.lookpolymers.com/pdf/Covestro-Bayblend-FR-110-PolycarbonateABS-Blend.pdf
[3] https://www.sabic.com/en/products/polymers/polycarbonate-pc-polybutylene-terephthalate-pbt/xenoy-resin
[4] https://encompolymers.com/
[5] M. Sepe: Dynamic Mechanical Analysis for Plastics Engineering, Elsevier
[6] https://www.findoutaboutplastics.com/2022/11/polymer-material-selection-how-can-flow.html